Update time:2026-03-17Visits:3985
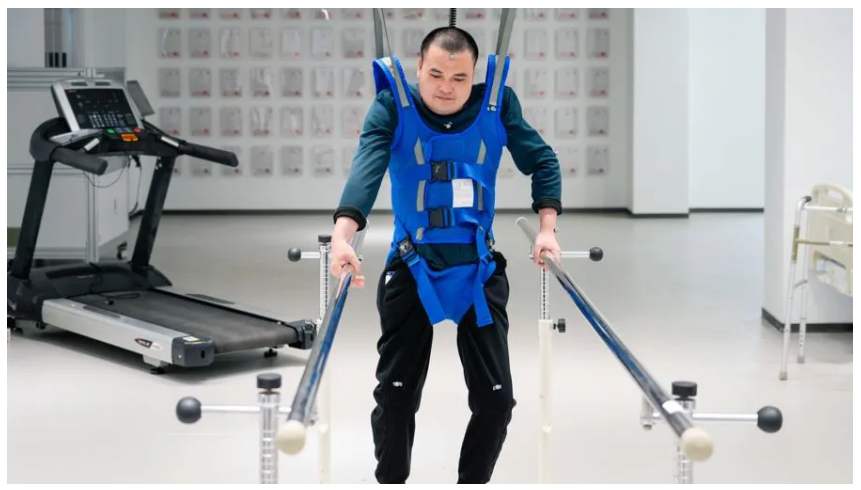
Picking up a glass of water, holding a grape, writing the words “thank you”—these simple, everyday actions were once impossible for patients paralysed by the loss of hand movement.
Now, a groundbreaking brain-computer interface (BCI) technology developed in Shanghai has turned these fundamental human desires into reality. This system is more than a mechanical link; it is a bridge that reconnects individuals to the world.
Today, the implantable BCI Hand Movement Function Compensation System—named NEO—developed by Boréas Medical Technology (Shanghai) Co., Ltd., has become the world’s first invasive Class III medical device of its kind to receive full market approval.

Why Shanghai?
Behind this global first lies not only the relentless drive of one company, but also Shanghai’s strategic ambition to establish itself as the "Global Innovation Source and First Launch Site for Brain-Computer Interfaces." It reflects the city’s deep commitment to nurturing future industries through a comprehensive, end-to-end innovation ecosystem.
Venturing into the Unknown
For Boréas founder Xu Honglai, his earliest and most vivid impression of BCI dates back to his freshman year in a Neural Engineering Laboratory at Tsinghua University. There, he saw a researcher who had shaved his head for an experiment. The lab was filled with imported equipment, and even the initial research data came from overseas.
That moment planted a seed. From its founding in 2011, Boréas focused on the core research and industrial application of BCI technology, making it one of China’s earliest dedicated BCI companies. After five years, it developed the world’s fastest EEG signal acquisition module in terms of communication rate, creating a vital tool for domestic neuroscience research.
At the time, non-invasive BCI products were already in use at several hospitals. Boréas faced a critical choice: follow the established path or venture into the technological unknown? Non-invasive technology, with electrodes placed on the scalp, is inherently limited by signal quality. Could they go deeper—placing electrodes inside the skull? After engaging directly with patients, Boréas committed to the more challenging path of epidural implantation. The team agreed: the true value of BCI lies in solving urgent clinical needs, not in technological spectacle.
The "unknown" proved far more difficult than anticipated. Electrode biocompatibility, long-term implant stability, minimising surgical trauma—each hurdle had no precedent, and every breakthrough meant pushing the boundaries of the possible.
Clinical trials were an even greater challenge. In October 2023, the NEO system was first implanted in a patient at Xuanwu Hospital, Capital Medical University. By August 2024, it had entered the China National Medical Products Administration’s special review channel for innovative devices. Starting in May 2025, multi-centre clinical trials launched across 11 hospitals nationwide, setting an industry record by completing 32 cases in just 78 days.
Professor Mao Ying, President of Fudan University’s Huashan Hospital, reported a 100% improvement rate in patients’ grasping function, with some even showing signs of neural rewiring. “We must not underestimate the act of grasping,” she noted. “It is profoundly important. For these patients, it is the first step back to self-reliance.”
Following implantation with Boréas’s brain-computer interface, paralysed patients are able to grasp a water bottle and drink unaided.
Securing certification is only the first step. “Our goal this year is to achieve the first clinical application in a patient,” the company states. “While ensuring quality, we will drive down manufacturing costs so the technology becomes accessible, beneficial, and affordable for patients.”

A Hundred Flowers Bloom
Boréas’s achievement is not an isolated one.
One entrepreneur describes Shanghai’s brain-computer interface (BCI) ecosystem as “streaking ahead of the pack.” As China’s first future industrial cluster for BCIs, the Shanghai Brain-Intelligence Hub brings together enterprises, leading hospitals, and research platforms, with nearly 30 dedicated BCI companies now registered. “The upstream and downstream supply chain can literally be found upstairs and downstairs in the same building,” they note.
Shanghai Brain-Intelligence Hub: China’s First BCI Future Industrial Cluster
The field of brain-computer interfaces faces a well-known “trilemma”—three core challenges that are difficult to optimise simultaneously: minimal invasiveness, long-term system stability, and high information throughput. The technical difficulty lies in the inherent trade-offs between them. In Shanghai, distinct technological pathways are already taking shape. The differentiated development of nearly 60 enterprises across the city has created a unique industrial landscape, establishing Shanghai as a pioneering global hub for innovation across every major BCI technology route.
By 2025, Shanghai had three invasive BCI products accepted into China’s Special Review Program for Innovative Medical Devices, with one invasive product also entering the U.S. FDA’s “Breakthrough Device” pathway. Multiple non-invasive products are already being applied in areas such as post-stroke motor rehabilitation, screening for depression and anxiety, and insomnia diagnosis. Market-ready products from companies like Surreal, Siyi, Aoyi, Quanlan, and Niantong are accelerating their commercial rollout.
Meanwhile, in Step Medical’s laboratory, the world’s smallest and most flexible neural electrodes are undergoing further refinement. Last year, in collaboration with the Chinese Academy of Sciences’ Center for Excellence in Brain Science and Intelligence Technology, Step Medical conducted China’s first invasive BCI clinical trial—an achievement listed among China’s top ten science and technology news stories of 2025. Unlike Boréas’s epidural approach, Step Medical’s technique inserts ultra-flexible electrodes directly into brain tissue to record signals. The typical human reaction time to an external stimulus is around 200 milliseconds. Step Medical’s team has compressed their system’s latency to under 100 milliseconds—lower than the body’s own physiological delay—effectively achieving “thought as action.” Their minimally traumatic invasive BCI system has now entered the “green channel” of China’s medical device review process for innovative products. “The future goal is to make implantation as simple as getting an ear piercing,” explains founder Li Xue. This July, Step Medical’s MEMS production base will commence operations, enabling the mass production of these advanced neural electrodes.
One month after surgery, the participant in China’s first invasive BCI clinical trial controls a racing game using only brain signals.
A patient with a tumour near the brain’s language centre imagined a motion, and a robotic hand responded by signing “Happy New Year 2025.” Real-time decoding of Chinese is a recognised major challenge in brain-computer interface (BCI) technology. This world-first prospective clinical trial for real-time Chinese decoding, conducted by NeuroXess, was the subject of in-depth reports on the official websites of Science and Nature. Months earlier, NeuroXess founder Tao Hu lost a video game match to a paraplegic patient who was completely paralysed from the shoulders down—the very same patient enrolled in China’s first clinical trial of a fully implanted, wireless, and fully functional BCI product. NeuroXess innovatively placed the battery module within the patient’s chest subcutaneous tissue, an area more tolerant of temperature changes, significantly improving system safety.

World-First Prospective Clinical Trial for Real-Time Chinese Decoding
Last Lunar New Year, paraplegic patient Xiao Lin felt a joy akin to rebirth—after undergoing brain-spine interface surgery, he was able to walk again. “On the first day after surgery, when I saw Xiao Lin lift his leg in bed, my anxious heart finally settled. I couldn’t help but share the news with everyone late that night,” recalled Jia Fumin, founder of NeuroHeal. As a form of generalised brain-computer interface, the brain-spine interface uses brain signals to regulate spinal nerves. At the end of last year, NeuroHeal’s minimally invasive brain-spine interface product—the first domestic medical device of its kind in China—entered the U.S. Food and Drug Administration’s “Breakthrough Therapy” approval pathway. To date, Jia Fumin’s team has completed the first batch of four nationwide clinical proof-of-concept cases. The trial’s paraplegic patients regained voluntary control of leg movement within 24 hours post-surgery, with the fastest achieving weight-supported walking in just 14 days.
The Project Managers
“We originally prepared for a protracted battle of at least 10 years, but we didn’t expect it to take just over six years from project launch to regulatory approval for market entry—far exceeding our expectations for speed,” said Xu Honglai.
In interviews, multiple companies frequently mentioned the “highly knowledgeable” project managers specialising in brain-computer interfaces. This key team, promoted by the Shanghai Municipal Science and Technology Commission, drives Shanghai’s BCI future industry initiative. Companies can communicate their needs to project managers by phone at any time. They often remain on-site solving problems until 10 p.m., and during critical phases, meetings are almost daily—a level of engagement they describe as being “embedded.”
To accelerate the clinical trial process for Boréas’s product, the project manager team collaborated with Huashan Hospital to coordinate 11 hospitals nationwide, creating a detailed timeline that compressed the originally planned one-year-plus trial cycle to just 78 days. “The company’s original workflow was like a series circuit; we helped them reconfigure it into a parallel circuit, dramatically improving efficiency. Daily schedules were broken down into specific time slots, advancing with military precision.”
When they learned that Step Medical was exploring a new vascular intervention pathway, the project managers quickly connected the medical device company with Huashan Hospital, establishing a cooperative platform. “In this process, it wasn’t us chasing them, but them frequently urging us on,” said Li Xue.
During company visits, the project managers identified that a key component—the feedthrough filter—was primarily imported and in high demand across almost all invasive BCI companies. They conducted a comprehensive market scan, coordinated innovation resources, and matched prototype products to meet this shared need.
“Connecting resources and tackling real-world challenges—this level of support was once unimaginable,” reflected Xu Honglai.
Shanghai’s recently launched future-industry project manager system represents a pioneering approach in China—one that maintains strategic agility in truly uncharted territory. “The defining feature of future industries is that their technological pathways are still unfocused and carry enormous uncertainty. We can’t afford to remain trapped in the old management model of issuing guidelines and evaluating projects based on past frameworks,” explained an official from the Municipal Science and Technology Commission. Since June 2024, the Commission has appointed a number of project managers dedicated to sectors such as brain-computer interfaces, quantum computing, green fuels, controlled nuclear fusion, 6G, and fourth-generation semiconductors. Their role is to oversee full-chain planning, end-to-end management, and lifecycle cultivation of these emerging fields.

The All-Rounder
When describing Shanghai’s brain-computer interface ecosystem, several companies independently used the same term: an “all-rounder.”
Their preference for Shanghai is no accident. In 2025, the city’s biomedical sector defied broader trends to surpass the trillion-yuan threshold for the first time, with manufacturing output nearing 210 billion yuan—a year-on-year increase of 2.4%. If Elon Musk’s brain-computer interface ventures operate within a closed, proprietary system, Shanghai’s approach resembles an open-source platform. Thanks to its industrial scale, ecosystem foundations, and global outlook, the city has developed a uniquely advantageous “interface” capability.
In building its brain-computer interface industry, Shanghai has long moved beyond supporting individual companies or projects. Instead, it has fostered a deeply integrated ecosystem encompassing government, industry, academia, research, finance, application, and services. Since 2017, the city has launched a series of major municipal-level initiatives in brain science and brain-computer interfaces, supporting breakthroughs in core technologies like flexible electrodes and accelerating later-stage product development. This environment has nurtured the rise of local leaders such as Step Medical and NeuroXess. “Shanghai’s brain project was launched three years ahead of the national brain initiative,” recalled Mao Ying. At the time, building a brain database urgently required specialised talent—and despite complex recruitment procedures, Shanghai had the vision to open a green channel.
The city’s industrial policy has consistently adhered to a core logic of “product-led, clinically applied.” In January 2025, it issued the Shanghai Brain-Computer Interface Future Industry Cultivation Action Plan (2025–2030)—the first provincial-level action plan of its kind in China, explicitly emphasising “medical-grade scenarios as the core and strategic products as the focus.” It is precisely this philosophy that has allowed Shanghai to seize the initiative in commercialising brain-computer interface technology. Policy coordination has been equally critical. The city has established a cultivation mechanism built around project-manager teams taking the lead, key-task lists driving breakthroughs, future-industry funds enabling growth, and future-industry clusters providing support. This creates cross-departmental and cross-regional synergy, helping companies avoid unnecessary detours on their path to industrialisation.
As a leading hub for brain-computer interface (BCI) research in China, Shanghai’s Lingang Laboratory is spearheading a national innovation consortium. Meanwhile, Fudan University has developed the world’s first whole-brain-scale simulation platform, capable of modelling 86 billion neurons. Shanghai Jiao Tong University’s SEED emotional EEG dataset is now used by more than 800 institutions worldwide. These efforts reflect Shanghai’s deepening commitment to foundational BCI technologies, where a steady stream of original breakthroughs is injecting fresh vitality into industrial innovation—and gaining global attention. In recognition, the prestigious International Brain-Computer Interface Conference will hold its first Asian session in Shanghai in 2024.
Xu Honglai once faced the challenge of intracranial sensor electrodes shifting position, where even a slight displacement could distort signal acquisition. Doctors ultimately resolved this by suturing the electrodes outside the dura mater. “They said it came down to the fundamental skill of suturing something as delicate as grape skin,” he remarked. The industrialisation of BCI relies on top-tier clinical expertise, an area where Shanghai excels. As the National Center for Neurological Diseases, Huashan Hospital maintains nearly 1,000 neurosurgery beds and performs over 20,000 brain surgeries annually. It was also the first medical institution in China to achieve full clinical implantation of a BCI. The hospital’s iBRAIN invasive EEG data alliance has gathered high-quality data from more than 500 patients, supporting ongoing algorithm optimisation. “We accurately located the hand motor and sensory brain area in just 3.3 minutes,” explained Professor Mao Ying. During the first clinical trial in Shanghai for Boréas’s BCI products, Huashan Hospital employed millimetre-level precision positioning technology, dramatically reducing surgical time.
Regulating BCI technology is itself an innovative endeavour, requiring authorities to first deeply understand the field. Shanghai is taking the lead in constructing a regulatory support system tailored to this cutting-edge discipline. The Shanghai Medical Device Testing Institute conducts over 90% of the type testing for implantable BCI products nationwide and has pioneered China’s first BCI medical device industry standard. The city has also established a dedicated ethics committee for BCI clinical trials.
In 2024, amid a broader biopharmaceutical downturn, securing financing became exceptionally difficult for companies. Xu Honglai acknowledged that at the time, Boréas had completed only two clinical trials. A critical round of funding was then led by Futeng Capital, under Shanghai Guotou, and Pudong Venture Capital. “This changed my perception of state-owned capital investment. I expected the process to be slow, but this cooperation showed me the ‘Shanghai speed’,” he said. Shanghai has built a multi-tiered investment and financing ecosystem, with a combined pool of industry funds totalling 47.5 billion yuan—including mother funds, future industry funds, and biopharmaceutical merger and acquisition funds—all working in concert to provide full lifecycle financial support for pioneering fields like BCI. Last year, BCI firms nationwide raised nearly 3 billion yuan, half of which was secured in Shanghai. The city led the country in both the number and value of financing deals, underscoring its substantial capital advantage.
This comprehensive ecosystem accelerates growth for companies on the ground. “In just five years since founding in Shanghai, Step Medical has maintained a steep growth trajectory, with its development pace continually rising,” noted Li Xue.
“Nuclear Explosion Point”
“In the early days, the very idea of a brain-computer interface industry was unimaginable. The technology was purely experimental, and research equipment often had to be built from scratch,” recalls Xu Honglai. Back then, the occasional industry gathering would draw only a handful of familiar faces. Compare that to just a few months ago, at the Brain-Computer Interface Conference in Shanghai: the venue was packed. The momentum today is on a completely different scale.
Brain-computer interface (BCI) is one of the most promising frontiers in future industries, with enormous market potential. Projections suggest the global market could reach $40 billion by 2030 and approach $150 billion by 2040.
With the launch of products like NEO, patients with spinal cord injuries will be among the first to benefit, while treatments for conditions such as epilepsy, stroke, and ALS are also accelerating. According to Shanghai’s industrial roadmap, by 2027, more than five invasive BCI products are expected to complete medical device certification and clinical trials, helping patients with aphasia or paralysis regain partial language and motor function. By 2030, the city aims to cultivate multiple industry leaders, striving to build a trillion-yuan-scale cluster and establish itself as a global hub for BCI innovation.
The central questions now: How can we unlock more applications for BCI? How can we achieve more seamless interaction between carbon-based life and silicon-based intelligence? Shanghai is forging consensus and pushing forward in exploration. The technology is like a “hammer”—the challenge is to find the right “nail.” And the current industry boom is opening up limitless possibilities for BCI’s diversified development.
Brain-computer interface is not only redefining our relationship with disease; it is gradually turning thought into productivity and expanding the boundaries of human capability. Technology, after all, exists to serve people. Shanghai’s innovative drive aims not only to restore hope to patients but also to catalyze industrial upgrading, create a “tipping point,” and ensure this transformative technology benefits society at large.
That the world’s first approved Class III invasive BCI medical device emerged in Shanghai is no accident. Through full-chain support, breakthroughs in innovation mechanisms, and a collaborative ecosystem, the city has built a distinctive edge for the industry—answering the question, “Why Shanghai?”
As one of the world’s most significant hubs for BCI, Shanghai now stands on the brink of an industry breakthrough. The coming two years may well mark BCI’s “DeepSeek” moment.
In this city, alive with innovative energy and imagination, the story of brain-computer interfaces is only just beginning. The future holds extraordinary promise.
The World’s First Invasive Brain-Computer Interface Just Got Approved in Shanghai
A Shanghai Hospital Launches Vertical Large Model for Women’s Health
Shanghai Research Team Develops AI Diagnostic System for Rare Diseases with “Explainable” Reasoning
“Yi Jian Kang” Foundation Established by Professor Wen Yumei Celebrates 13 Years of Impact
Shanghai Hosts Landmark Global Transplant Congress, a First for Mainland China

The World’s First Invasive Brain-Computer Interface Just Got Approved in Shanghai

A Shanghai Hospital Launches Vertical Large Model for Women’s Health

Shanghai Research Team Develops AI Diagnostic System for Rare Diseases with “Explainable” Reasoning

World’s First “Video Camera” MRI is set in Shanghai, Ushering in the Era of Dynamic Physiological Imaging

“Yi Jian Kang” Foundation Established by Professor Wen Yumei Celebrates 13 Years of Impact

Renji International Medical Marks 20th Anniversary

Shanghai Overseas Patients See 25% Growth

Shanghai Hosts Landmark Global Transplant Congress, a First for Mainland China

Shanghai Health Report, Issue 7: Expert Guidance

Shanghai Doctors Become Viral Health Educators